Diagram representation of the element sulfur Vector Image
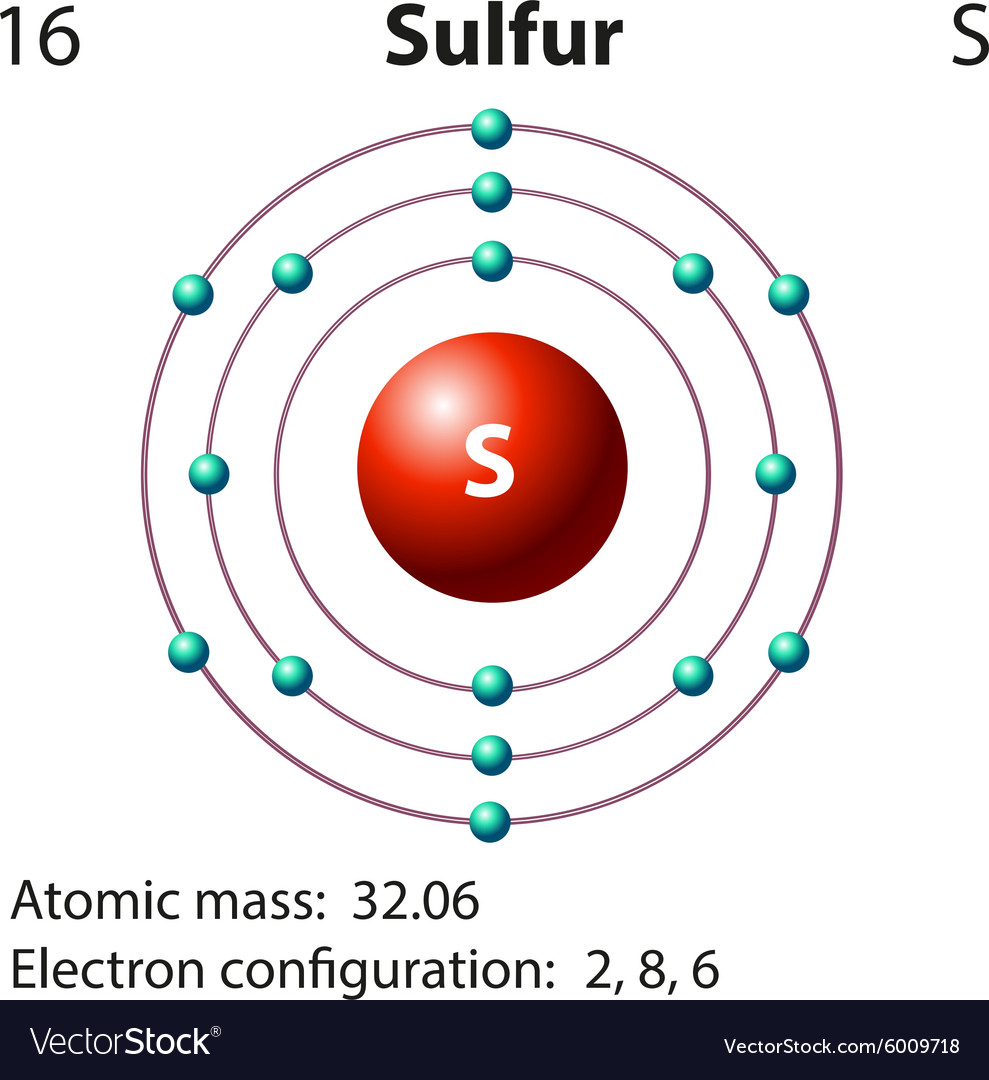
Drawing the Bohr Model of Sulfur. Sulfur is the 16 th element of the Periodic table. It belongs to Period 3 and group 16. The information that we can infer from the above-mentioned Sulfur box is as follows: • The atomic number of Sulfur is 16. • The electronic configuration of Sulfur is [Ne] 3s 2 3p 4.
Periodic Table Sulfur Atom Periodic Table Timeline
In this video we'll look at the atomic structure and Bohr model for the Sulfur atom (S). We'll use a Bohr diagram to visually represent where the electrons a.

Sulfur Bohr Model — Diagram, Steps to Draw Techiescientist
For example, sulfur has an atomic number of 16, so its Bohr diagram would show two electrons in the first energy level and eight electrons in the second energy level. The Bohr diagram provides a visual representation of an element's electron arrangement, making it easier to understand and analyze the behavior of atoms.
Sulfur Table of Elements by Shrenil Sharma
Bohr Diagram: The First Element. In order to make a Bohr diagram, you need to know the number of protons, neutrons, and electrons the element has. In this section, we'll show a sample Bohr diagram for hydrogen. H —Hydrogen. 1 proton. 1 electron. 0 neutrons

Sulfur Atom Science Notes and Projects
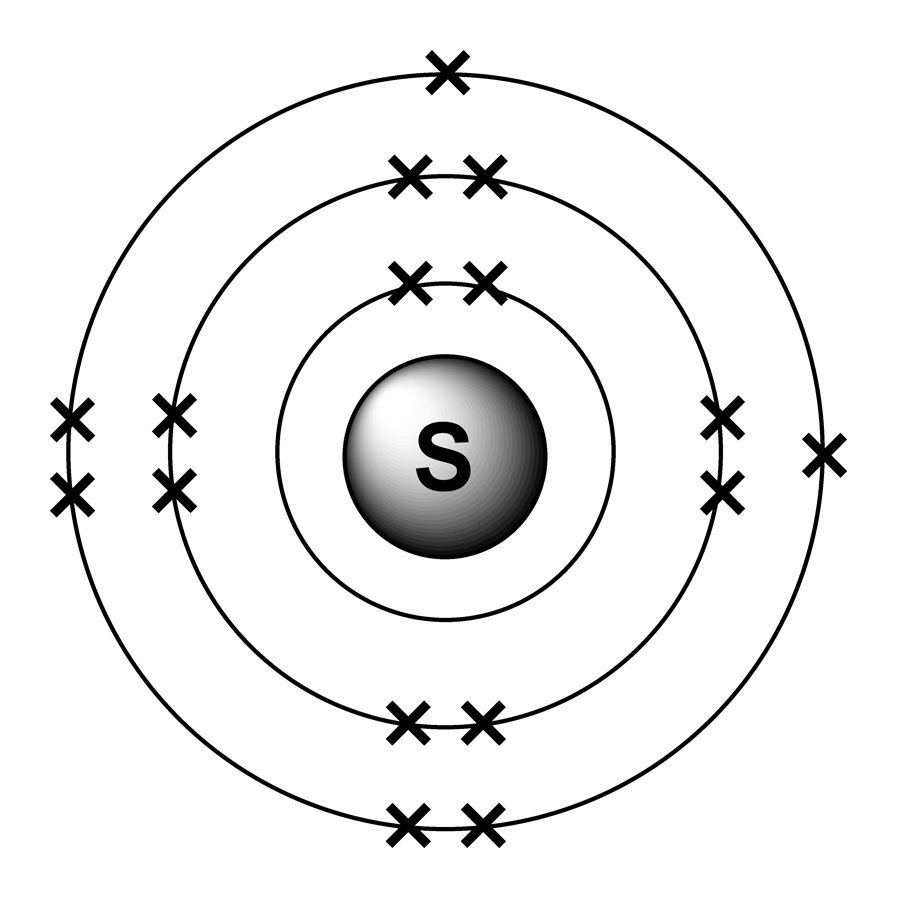
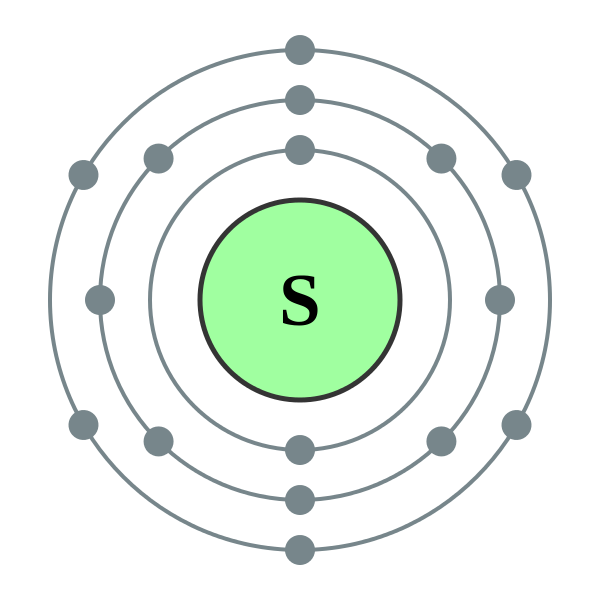
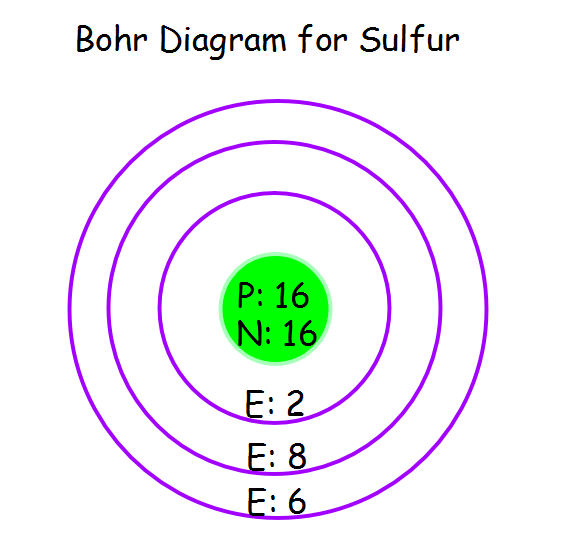
The Bohr model of Sulfur (S) is drawn with three electron shells, the first shell contains 2 electrons, the second shell contains 8 electrons and the third shell contains 6 electrons. Sulfur is neutral and its atomic number is 16, hence, the number of protons and electrons available for its Bohr diagram is also 16.

Sulfur atom tewswheel
Bohr model of Elements. 1. Hydrogen (H) 1. 2. Helium (He) 2. 3. Lithium (Li)

Bohr Diagram Of Sulfur
The Bohr Rutherford diagram of sulfur illustrates that it has 16 electrons distributed in different energy levels or shells around the nucleus. The first energy level contains two electrons, and the second energy level contains eight electrons. The outermost or valence shell contains six electrons.

Bohr Diagram Of Sulfur Wiring Diagram
Sulfur / Sulphur has 2 electrons in its first shell, 8 in its second, 6 in its third.Check me out: http://www.chemistnate.com
Sulfur Bohr Model Diagram My XXX Hot Girl
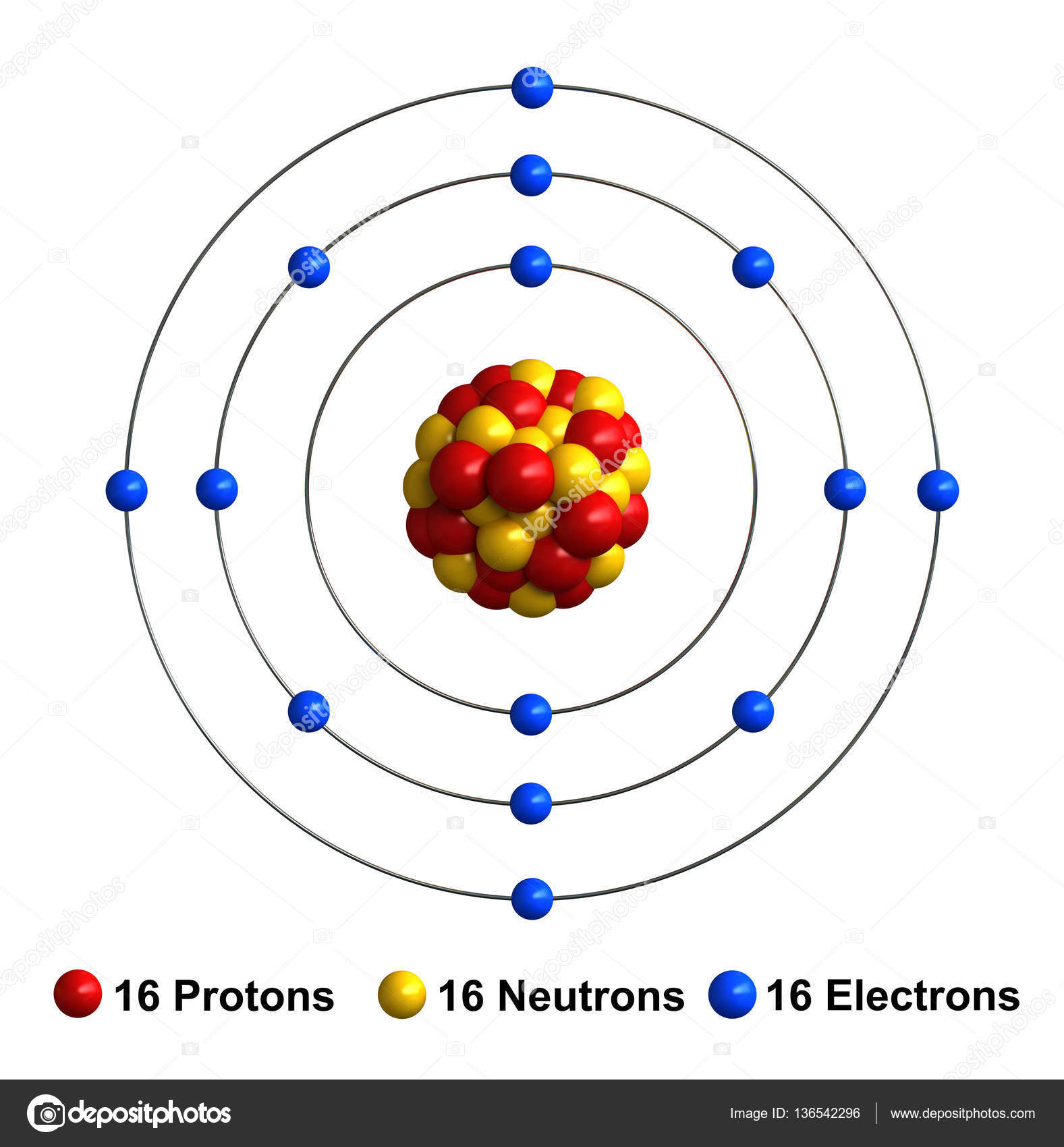
The sulfur atom has 16 protons, 16 neutrons and 16 electrons in three different energy levels, or orbits. Physics suggests that electrons do not physically exist as "points," but teachers use the Bohr atom model with fixed electrons as a way to simplify atomic structure. Creating the model requires the ability to cut with scissors and use glue.
Bohr Diagram Of Sulfur
Bohr model of calcium: (CC BY-SA 2.0 uk;Greg Robson): Answer b. Bohr model of sulfur: (CC BY-SA 2.0 uk; Greg Robson). Valence electrons are located in the highest energy level of an atom. When drawing a Bohr diagram, the valence electrons would be present in the outermost electronic level/shell (furthest away from the nucleus).
Bohr Diagram Of Sulfur
The atomic number of sulfur represents the total number of electrons of sulfur. Since the atomic number of sulfur is 16, the total electrons of sulfur are 16. Calculate the maximum number of electrons each subshell can hold using the formula: 4ℓ + 2. Finally, use aufbau chart and start writing electron configuration.
:max_bytes(150000):strip_icc()/sulfuratom-58b602563df78cdcd83d5a9d.jpg)
Atom Diagrams Electron Configurations of the Elements
That is, sulfur is an anion element. S + 2e - → S 2-. The electron configuration of sulfide ion (S 2-) is 1s 2 2s 2 2p 6 3s 2 3p 6. This electron configuration shows that sulfide ion (S 2-) has three shells and the 3rd shell has eight electrons.

Sulfur Bohr Model — Diagram, Steps to Draw Techiescientist
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is.
Bohr Diagram The Element Sulfur
What is the Bohr diagram for sulfur? Bohr Model: In the Bohr model, electrons are confined to concentric spheres around the nucleus numbered as n=1, 2, 3,.. The sphere n = 1 can accommodate two, the n=2-3 spheres eight, and the n = 4 sphere fourteen electrons. Answer and Explanation: 1.
Electron Configuration Noble Gas Atom Bohr Model Chemistry Sulfur
The Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jumps between orbits, is accompanied by an emitted or absorbed amount of electromagnetic energy (hν). The orbits in which the electron may travel are shown as grey circles; their.

Sulfur Bohr Model — Diagram, Steps to Draw Techiescientist
Here's how you can draw the Bohr model of sulfur step by step. #1 Write protons, neutrons, and electrons of sulfur atom. #2 Draw nucleus of sulfur atom. #3 Draw 1 st electron shell. #4 Draw 2 nd electron shell. #5 Draw 3 rd electron shell. Let's break down each step in detail.